Computational modeling (CM) is the use of computers to build a model of a plan or procedure to find ways to improve outcomes. Early research indicates that studying this type of modeling may have a large impact on otolaryngology surgery at all stages.
The hope is that CM will eventually be able to guide surgeons to help prevent potential errors, increase patient safety, and predict outcomes. The information would be based on known scientific evidence indicating that, if a procedure is performed a certain way, the physician can expect certain types of outcomes, said Grace C.Y. Peng, PhD, a program director in the division of discovery science and technology at the National Institute for Biomedical Imaging and Bioengineering (NIBIB) in Bethesda, Md. “The idea is to eventually develop models to be more predictive and more proactive to get the best possible outcome on the first try,” she added.
Surgery is a common treatment for nasal airway obstruction (NAO); however, its failure rate is high. “Currently, between 25% and 50% of surgeries to correct NAO fail to completely resolve the patient’s symptoms,” said Dennis O. Frank-Ito, PhD, an assistant professor of surgery in the division of head and neck surgery and communication sciences at Duke University Medical Center in Durham, N.C. “This implies that as many as one out of every two surgeries will not produce the desired outcome,” he added. For this reason, he and his colleagues became interested in determining how they could apply computational technologies to aid surgical planning for NAO treatment, potentially improving outcomes.
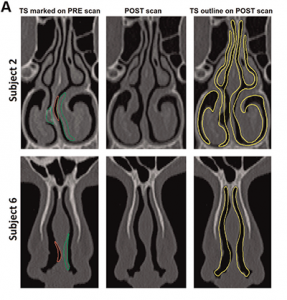
Figure 1. 3D transcribed-surgery models. Left: Digitally created airspace (green) and closed airspace (red). Right: Transcribed-surgery (yellow) superimposed on postsurgery.
© marcinswiostek / shutterstock.com
As an early proof-of-concept study, Dr. Frank-Ito and his team prospectively constructed nasal models from computerized tomography (CT) scans of 10 patients undergoing surgical treatment for NOA (see Figure 1, and Figure 2). The scans were taken prior to surgery, and five and eight months following surgery. To create the transcribed-surgery and post surgery models, the surgeon digitally hand-edited the preoperative scans to represent the physical changes that were expected from the surgery. Steady-state, laminar, and inspiratory inflow were simulated in each model. The results were not as robust as they had hoped, largely related to software limitations (Otolaryngol Head Neck Surg. 2014;151:751-759).
“We hope that, eventually, this will become a way for the surgeon to meld radiologic and other studies into a platform that lets them virtually plan and even practice long before they enter the nose,” said Dr. Frank-Ito. “The pre-surgery platform would allow surgeons to ‘try out’ different procedures virtually before deciding the best procedure to perform on a given patient.”
Each virtual procedure tried on the CM platform should produce an expected patient improvement level. Dr. Frank-Ito thinks this will lead to personalized and optimal surgical treatment for patients.
With further development, more advanced tools could be developed, providing real-time guidance for physicians and, hopefully, making surgery easier and more successful. —Jason Lu, PhD
Predicting Patient-Specific Outcomes
Another possible use of CM is to predict individual postoperative complication rates. One such program looked at forecasting adverse outcomes following oral cavity cancer surgery.
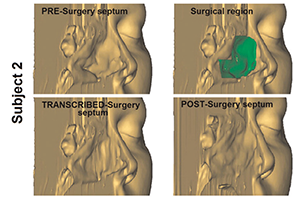
Figure 2. 3D transcribed-surgery model of the nasal septum.
“Head and neck cancers are complex, particularly in the surgical management,” said Mahmoud I. Awad, MD, resident physician in the head and neck service of the department of surgery at Memorial Sloan Kettering Cancer Center in New York City. “We find it is very difficult to predict postoperative complications and physicians often just use their clinical judgment.” This can be frustrating, added Dr. Awad. So, he and his colleagues set out to develop nomograms to better quantify this risk, and to be able to relay the risk to patients during presurgical counseling (JAMA Otolaryngol Head Neck Surg. 2015;141:960–968).
The researchers retrospectively reviewed the charts of 506 patients treated surgically for primary oral cavity squamous cell cancer at their institution. One cohort was used to develop nomograms examining 36 potential complication predictors and to determine which were statistically more likely to predict complications. From this information, the group then constructed the nomograms. The other cohort was used to validate the nomograms once they were designed.
Clinical characteristics were similar between the groups for most comparisons. The six preoperative variables with the highest individual predictive values were incorporated into the nomograms. The nomogram predicted major complications with a validated concordance index of 0.79. When they added surgical operative values, predictive accuracy was maintained at 0.77.
“What is great about nomograms is that they are a combination of being user friendly and allowing us to personalize medicine,” said Dr. Awad. “All the variables entered into consideration are specific to the individual in front of you. In theory, the information you are giving them on risk is specific to that individual patient. It is a very powerful tool.”
Dr. Awad is confident that this method has the possibility to change practice significantly. The clinician should be able to enter variables into a computer in real time and get results to share with the patient immediately. In addition, being able to assign a specific risk to a patient could have implications in other areas of healthcare, such as allocation of intensive care unit beds and risk adjustment to normalize outcomes among different facilities for quality and pay-for-performance purposes.
We hope that eventually, [CM] will become a way for the surgeon to meld radiologic and other studies into a platform that lets them virtually plan and even practice long before they enter the nose. —Dennis O. Frank-Ito, PhD
Assessing CI Success
One concern physicians have about the use of cochlear implants (CI) in prelingual infants and toddlers is the difficulty of predicting the development of effective language skills in the first two years following surgery. Researchers from Cincinnati Children’s Hospital Medical Center and the University of Cincinnati developed a computer-based machine learning model to try to address this issue (Brain Behav. 2015 Oct 12;5:e00391. doi: 10.1002/brb3.391).
Jason Lu, PhD, associate professor in the division of biomedical informatics at Cincinnati Children’s, and his colleagues analyzed results of pre-surgery hearing tests. The researchers found that elevated activity during functional magnetic resonance imaging (fMRI) in two regions of the brain was highly predictive of which child would benefit the most from their implants.
The stimuli used in the fMRI testing before the surgery included both natural language speech and narrow-band noise tones. When the investigators analyzed data from both pre-surgery auditory tests and two-year language tests, the brain activation patterns in natural language speech showed greater predictive ability. “This study identifies two features of our computer analysis that are potential biomarkers for predicting CI outcomes,” said Dr. Lu. “More accurate screening could reduce the number of children undergoing this costly and invasive procedure, only to be disappointed with the results.”
Early Stages Still
Although CM is promising, the researchers stressed that the tool is still very much in the proof-of-concept stage. The studies have been prospective in nature and small in number. They are also looking at the next stage of development, which will include using larger datasets and validating the results in other institutions.
“CM is a very powerful and promising tool, but it requires a close collaboration between physicians and computational scientists to develop a model,” said Dr. Lu. “The physicians provide domain knowledge to computational scientists. With further development, more advanced tools could be developed, providing real-time guidance for physicians and, hopefully, making surgery easier and more successful.”
Surgeon/Programmer Communication Essential
Better communication and understanding among the players involved may help build trust in the models. “One of the reasons there is skepticism around the use of models is that people don’t know what is under the hood,” said Dr. Peng. “They don’t have a good idea of how the numbers were found and what they mean.” One of her bigger challenges is getting modelers in contact with potential users, she said. “More people could then embrace modeling in their research and clinical practices. This would hopefully lead to models that are usable at the bedside independent of the size of the facility,” she added.
The next step is trialing these CM algorithms in a more structured manner. After that, there is confidence that the computer/clinical interface will become more important as times goes on. “I am hoping within the next 10 years we’ll have an initial set of surgeons embracing this technology once it becomes available,” said Dr. Frank-Ito. “By integrating CM with surgical navigation systems, doctors will be able to plan and even practice different procedures long before they enter the operating room. With the aid of navigation systems, our CM approach will identify sites of greatest nasal obstruction and then guide surgeons to these sites for correction. Our goal is that the surgeon will use our technology to complement what is currently done, rather than replace what they do.”
Kurt Ullman is a freelance medical writer based in Indiana.
Key Points
- The hope is that CM will be able to guide surgeons to help prevent potential errors, increase patient safety, and predict outcomes in head and neck surgeries.
- CM could also be used to predict individual postoperative complication rates.
- Although promising, CM is still in the proof-of-concept stage, and studies have been prospective in nature and small in number.
Computer Modeling in Medical Research
Computational modeling (CM) is becoming an important part of medical research. This is especially true in the surgical specialties.
The NIBIB was established in December 2000 and focuses its work on integrating engineering with physical and life sciences. The institute funds more than 700 grants and the work of approximately 5,000 researchers around the country and internationally. “[The NIBIB is] working with researchers who are looking at computer models to not only systematically guide therapy, but also plan surgery and predict outcomes,” said Dr. Peng.
One of the areas under intensive research is the development and use of computer models for health research and clinical practice. “With modeling, you have the beauty of utilizing a platform where you can try an infinite number of scenarios that you can’t try in real life,” said Dr. Peng. “This helps you see what would be the optimal way to do the surgery or to have certain outcomes occur.”
Many of the models are still basic science, however. For example, recipients of one NIBIB grant are studying nasal surgery parameters. Currently, information is lacking on which biophysical variables best predict symptom changes, what ranges of objective values are normal, and how best to make a virtual surgery tool that can be used to develop the “correct” surgical plan for each patient. The investigators are hoping to better understand which indicators are important and meld them into a computer model that would help address these issues during pre-surgical planning.
Postoperative Applications
There is also developing evidence that CM will have an application in the postoperative world. A program could use patient-specific clinical information and in-surgery indicators to help develop risk measurements for postoperative complications.
With this information, the surgeon and the rest of the team might be able to better manage the patient, by taking certain precautions, and the healthcare resources, by deciding who is most likely to need an intensive care unit bed and planning accordingly. “Think of this as a medical equivalent of one of the tax programs you or your accountant uses,” said Dr. Peng. “You put in some numbers, [along with] additional information like your address and Social Security number. The model under the hood then asks the right questions and does the calculations for us.”—KU