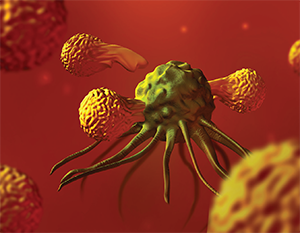
T lymphocyte cells (orange) surrounding a cancer cell.
© Spencer Sutton / Science Source.COM
This year, the National Institutes of Health estimates that approximately 60,000 Americans will be diagnosed with head and neck cancer. Although survival rates have increased over the last 30 years, the five-year relative survival rate for oral and oropharyngeal cancers is still only approximately 60% percent. Otolaryngologists, surgeons, and medical and radiation oncologists have had little to offer to patients whose disease has progressed despite surgery, chemotherapy, and radiation—until now.
Explore This Issue
September 2016In early August 2016, the U.S. Food and Drug Administration approved pembrolizumab (Keytruda, Merck Sharp and Dohme Corp.) for use in patients with recurrent or metastatic head and neck squamous cell carcinoma that is refractory to platinum-based chemotherapy. The drug received accelerated approval based on early data from the KEYNOTE-012 trial. Trial participants received 200 mg of pembrolizumab IV every three weeks for 24 months, or until disease progression or they reached intolerable toxicity. The overall objective response rate was 16%, with a complete response rate of 5%. The drug elicited response in both HPV-positive and HPV-negative tumors (24% vs. 16%). Eighty-two percent of those who responded to the medication did not have progression of disease for at least six months. Now, some patients are doing well more than two years out from their initial treatments (J Clin Oncol. 2016;34(suppl; abstr 6011)).
The traditional platform for treating head and neck has been a table with three legs, surgery, radiotherapy, and chemotherapy. Immunotherapy, in essence, brings a fourth leg to the table. —David Brizel, MD
Pembrolizumab is a checkpoint inhibitor, a drug that essentially unmasks tumor cells so the immune system can attack and destroy them. Specifically, pembrolizumab is a PD-L1 inhibitor; it inhibits the interaction of PD-L1, a protein on tumor cells and PD-1, a protein on T-cells. Checkpoint inhibitors “turn off the off switch, which has the net effect of turning the immune system back on,” said David Brizel, MD, Leonard Prosnitz Professor of Radiation Oncology at Duke University in Durham, N.C.
The idea of unleashing the immune system against cancer cells is not new. What is new is the hope that these medications are offering hope to patients who would otherwise have no options. Maura Gillison, MD, PhD, professor of medicine and Jeg Coughlin Chair of Cancer Research at The Ohio State University Comprehensive Cancer Center in Columbus, has seen remission following checkpoint inhibitor treatment in some of her patients.