Reprinted from Oncology Times, May 25, 2006.
Explore This Issue
July 2006New guidelines published by the American Thyroid Association hope to offer stronger evidence on which to base diagnosis and treatment of thyroid nodules and thyroid cancer.
Unlike the organization’s original guidelines, which were based mostly on expert opinion, these updated guidelines use an evidence-based approach that included a comprehensive review of the medical literature followed by recommendations for diagnosis and treatment based on the quality of the studies found.
“Every recommendation has a level of evidence that is put next to it that describes whether we recommend it or don’t recommend it based on the amount of evidence supporting it,” said David S. Cooper, MD, Director of the Division of Endocrinology at Sinai Hospital of Baltimore, who chaired the Task Force that wrote the guidelines and is the President-Elect of the American Thyroid Association.
“The older guidelines did not have any of this,” he added. “They were just a set of recommendations based on expert opinion.”
Evidence from well-designed, well-conducted studies that directly evaluated the effects on health outcomes was considered the highest quality evidence and rated an A (strongly recommends) or F (strongly recommends against).
One area that was rated an F in these new guidelines, and which represented a change from the 1996 guidelines, was the recommendation for follow-up care for patients with benign thyroid nodules.
“The guidelines now ‘strongly recommend against’ levothyroxine-suppression therapy for benign thyroid nodules, at least in the absence of nodule growth,” said Ronald Cohen, MD, Assistant Professor of Medicine in the Section of Endocrinology at the University of Chicago.
‘The guidelines do a nice job of identifying unresolved areas and differentiating the types of evidence on which recommendations are based.’
New Roles for Diagnostic Tools
The updated guidelines also provide new roles for diagnostic tools including recombinant human serum thyrotropin (rhTSH), ultrasound, and PET/CT scans.
For example, along with playing an important role in identifying recurrent disease in the neck of patients with established thyroid cancer, the updated guidelines also emphasize that ultrasound is helpful in the evaluation of thyroid nodules, to show that the palpable abnormality represents a thyroid nodule and to identify high-risk characteristic, Dr. Cohen noted.
Few Randomized Treatment Trials
In terms of therapy, however, the recommendations offered by the updated guidelines remain largely based on expert opinion, which carries a C (recommends) or D (recommends against) rating. This is largely due to the lack of randomized clinical trials, which provide the strongest evidence, on therapy options in thyroid cancer.
“I know in oncology it is unheard of to have a cancer and not have a randomized trial to figure out what to do about it,” said Dr. Cooper, who also is Professor of Medicine at Johns Hopkins University School of Medicine. “But in this particular situation, there really aren’t any.”
The reason for this, he explained, is that most patients with thyroid cancer have indolent, slow-growing disease that is cured simply by surgically removing the tumor. For destroying microscopic disease left behind by surgery, radioactive iodine is commonly given.
However, based on the evidence, use of radioactive iodine is not fully supported. Dr. Cooper said the evidence is fairly slim that adjuvant radioactive iodine is beneficial: “This is sort of a standard thing that people are getting, but we point out in the guidelines that it’s not necessarily proven beyond a shadow of a doubt that this is actually beneficial.”
For Dr. Cohen, the guidelines emphasize the need for better evidence on this issue. “Studies are needed to define the appropriate patients to treat with radioactive iodine, and the best doses of radioactive iodine to use,” he said.
Radioactive Iodine for Higher-Grade Tumors
Although no randomized evidence is available on which to recommend treatment, the guidelines do provide evidence that supports giving radioactive iodine in people with higher-grade tumors.
“In terms of what is the appropriate candidate for getting radioactive iodine, we now tie them to the TNM classification,” he said. “We admit that the level of evidence is not A-level evidence, but there is evidence supporting doing this in people with higher-grade tumors.”
Unresolved Areas and Types of Evidence
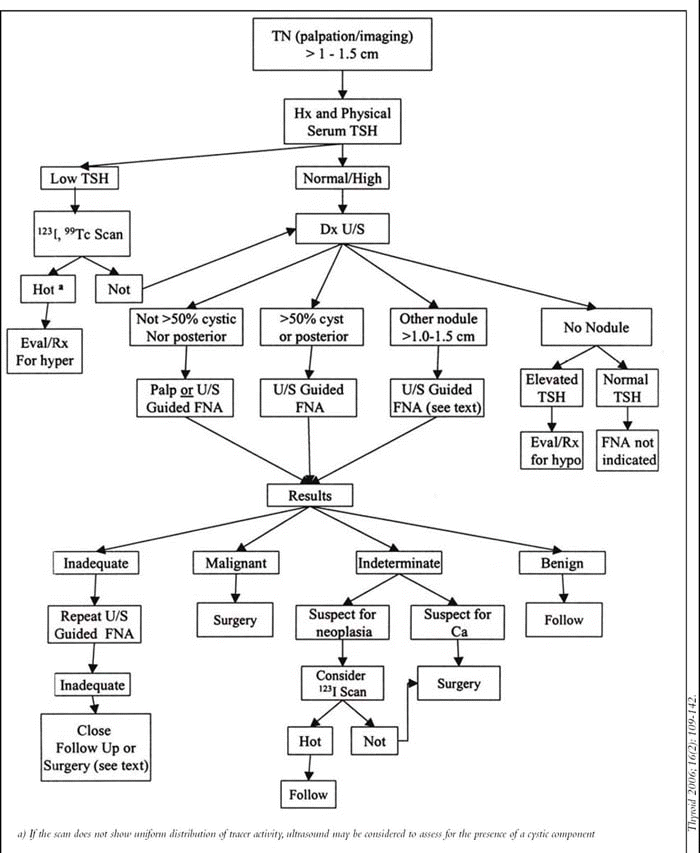
Dr. Cohen said the guidelines do a nice job of identifying unresolved areas and differentiating the types of evidence on which recommendations are based.
“The authors note when their recommendations are based on expert opinion as opposed to clinical trials, and when there is not enough information to even make a recommendations,” he said, adding that for unresolved issues treatment decisions need to be individualized.
Another area that remains unresolved and needs further research is more effective treatment for patients with advanced disease.
Unlike the American Thyroid Association’s original guidelines, which were based mostly on expert opinion, this updated version uses an evidence-based approach that included a comprehensive review of the literature followed by recommendations for diagnosis and treatment based on the quality of the studies found.
The final section of the guidelines is devoted to directions for future research and highlights novel therapies under investigation, particularly for patients with advanced thyroid cancer.
Dr. Cooper said that although patients with metastatic disease comprise a distinct minority of patients with thyroid cancer, new treatments for these patients are greatly needed given that no good treatment currently exists and that patients ultimately die of disease.
“Basically, there are a lot of drugs now becoming available in clinical trials for patients with advanced thyroid cancer,” he said.
©2006 The Triological Society
Leave a Reply