Recent studies indicate that the Bravo pH monitoring system from Medtronic may offer a safe and effective diagnostic tool for pediatric patients suffering from symptoms of gastroesophageal reflux disease (GERD). The visible catheters and discomfort associated with traditional diagnostic studies, along with the clinical setting required for some tests, often cause anxiety in adult patients, and can be downright frightening to a child. The Bravo system may offer a more comfortable and convenient testing option.
Explore This Issue
April 2006Diagnostic Gold Standard
Testing for the pH level is the gold standard for diagnosing GERD. Esophageal pH testing measures how often stomach acid flows into the lower esophagus, and the amount and degree of acidity during the test period.
Traditional pH testing involves placing a catheter through the patient’s nose and down the esophagus. This can cause the patient significant nasal and throat irritation, impairing the ability to maintain a normal diet. The catheter itself remains visible, causing many patients to alter their exercise and activity routines during the test period. These changes in the patient’s routine may provide false negative findings and adversely affect the reliability of the results.
The Bravo system may also provide data more representative of the patient’s day-to-day activities than traditional pH monitoring methods because patients are able to maintain a normal diet and activity level during testing.
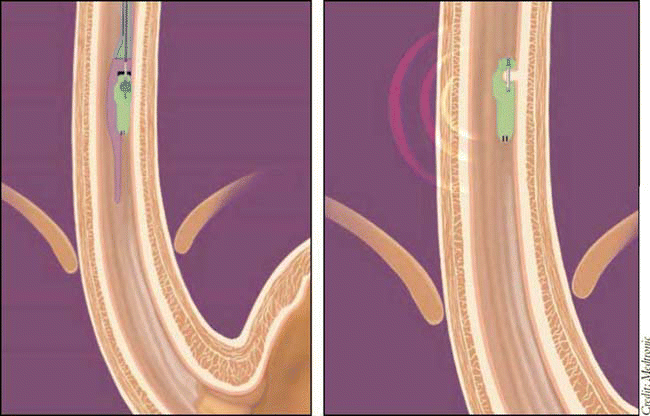
How the System Works
The Bravo system includes a capsule measuring 6 mm by 5.5 mm by 25 mm and containing a 3.5 mm deep well connected to an external vacuum unit. The physician inserts the capsule orally or transnasally into the esophagus during upper endoscopy. Using the vacuum unit, the physician pulls esophageal tissue into the well and literally pins the capsule into place. The delivery system is withdrawn, and the capsule is left in place to record pH data. Placement of the capsule takes about 10 minutes, and patient discomfort is minimal. Attachment of the capsule creates less trauma to the esophagus than a standard biopsy, and the point of attachment heals within several days.
Once the capsule is in place in the esophagus, it begins transmitting data via radio frequency telemetry to the Bravo receiver, which the patient wears on his or her belt. Once the test is complete, data are uploaded from the receiver to a computer for analysis and evaluation by a nurse or physician.

Leave a Reply