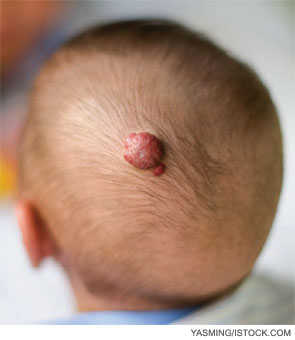
Since the first report in 2008 of the effectiveness of propranolol to treat infantile hemangiomas, its use has grown among physicians who treat these tumors, which arise in 5 to 10 percent of infants (N Engl J Med. 2008;358(24):2649-2651). Among these infants, approximately 10 percent will require treatment to correct functional impairment or prevent lasting cosmetic deformity caused by the hemangioma (N Engl J Med. 1999;341(3):173-181; Pediatr Dermatol. 1997;14(3):173-179).
The appeal of propranolol as a treatment choice is its demonstrated efficacy with reported lower side effect profile compared to other treatments, including corticosteroids, which have been the treatment of choice for many years.
To date, data are largely derived from small case studies and retrospective reviews. Propranolol appears to effectively halt growth of infantile hemangiomas and hasten involution with relatively rare side effects of sleepiness, night terrors, bradycardia, hypotension and hypoglycemia. A 2010 literature review of 11 case studies found that infants treated with propranolol for cutaneous, subglottic and oropharyngeal hemangiomas had significant regression, with minimal side effects (Int J Pediatr Otorhinolaryngol. 2010;74(4):338-342).
Other treatments, mainly systemic steroids, also seem effective, but the risk of side effects appears to be greater, particularly when used over time and in high doses. A study published in May reported that propranolol was effective for 26 hemangiomas of the nose, lips and parotid area, conditions researchers would normally have observed rather than treat with corticosteroid therapy (Arch Otolaryngol Head Neck Surg. 2011;137(5):471-478). Given the accumulating data on the efficacy and tolerability of propranolol in this setting, many otolaryngologists are adopting this agent as the first choice of treatment and even expanding the indications to treat these congenital anomalies.
“Propranolol appears to be an effective treatment for infantile hemangiomas and should now be used as a first-line treatment in hemangiomas when intervention is required,” said Annette Zimmerman, MD, assistant doctor of otolaryngology- head and neck surgery at Philipps-University Marburg and University Hospital Giessen and Marburg, in Marburg, Germany, and lead author of the literature review. Dr. Zimmerman emphasized propranolol’s well-documented safety and side effect profile in children based on more than 40 years of clinical use as therapy for infant cardiovascular disease.
Data Limited
Others, however, are voicing the need for more evidence to help physicians determine the true role of propranolol in this setting, given the lack of prospective data comparing the safety profile and efficacy of propranolol with corticosteroids. This call is particularly strong in the U.S., where propranolol is used off label.

Leave a Reply