Disease-specific quality of life (QoL) is improved with septorhinoplasty (SRPL), functional endoscopic sinus surgery (FESS), and combined surgery, and combining the procedures does not compromise QoL gain.
Hierarchical Task Analysis Can Help Reduce Error in FESS
HTA and the Systematic Human Error Reduction and Prediction Approach (SHERPA) are valuable tools to highlight potential errors in functional endoscopic sinus surgery (FESS).
Tasks Required for a Correct Training Approach to FESS
Ten tasks and 49 subtasks offer the opportunity to improve surgical training and enhance patient safety by identifying high-risk steps in functional endoscopic sinus surgery, and how risk can be mitigated.
Risks of Pharyngeal Pack Use Following FESS Outweigh Benefits
The authors advise against using the packs due to potential risks
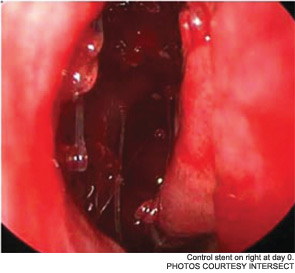
Drug-Eluting Sinus Stent Hits the Market: May help maintain patency after FESS
Maintaining sinus patency after functional endoscopic sinus surgery (FESS) for chronic rhinosinusitis (CRS) has long been an issue; as many as 23 to 47 percent of patients require revision surgery after FESS. Now, a new drug-eluting, bioabsorbable stent manufactured by Intersect (Palo Alto, Calif.) is being billed as a “breakthrough treatment [that] improves outcomes for sinus surgery,” according to a news release from the company. The device, which received pre-market approval from the U.S. Food and Drug Administration (FDA) in August, has been studied since 2008. It is currently available in Texas, New York, Philadelphia, New Jersey, Atlanta, Ohio and Kentucky.
Balloon Sinuplasty Use Continues to Evolve: Procedure may complement traditional sinus surgery
Six years after balloon sinuplasty was introduced to the otolaryngology community, it remains an evolving technology. “In my opinion, balloon dilatation has great potential, but it’s still trying to find its proper place in the ENT arena,” said Ralph Metson, MD, clinical professor of otology and laryngology at Harvard Medical School and Massachusetts Eye and Ear Infirmary in Boston.

Surgical Approaches to Sinus Disease: The Debate Continues
Part 2 of 2 articles
PHILADELPHIA-Surgeons are faced with many options for approaching diseases of the sinus; the right approach is not always clear-cut. Five experts reviewed several approaches at Rhinology World held here recently, with some favoring a more aggressive approach, some preferring to be less aggressive, and others highlighting new technology.